The Big Stuff-up 3
How to interpret reports of adverse events following Covid-19 vaccination
A serialisation of a letter to a relative in response to a publication from one of NZ’s leading Government mouthpieces- Stuff(ed) News.
The gist of this the Stuff article is that because anyone can make reports, they are probably inaccurate, or unrelated, and (without any evidence to back the claim up) over inflated. Missing from the article is any discussion of under-reporting of adverse events (AEs) in reporting systems like VAERS (USA), Yellow Card (UK), or CARM (NZ) which are well known and documented.
A rapid responsei supporting a British Medical Journal articleii on serious adverse events following the 2010 flu vaccine in Australia (where 1 in 110 children had a febrile seizure) Shows that the VAERS reported incidence of Kawasaki disease in children was 1 in a million whereas actual published data show a 40-80fold higher incidence.
This studyiii documents that the actual rate of confirmed severe anaphylaxis following C-19 Vaccination amongst 64,900 people in the study was 2.4 per 10,000. The reported rate in VAERS at the time was only 2.5 – 11.1 cases per million. So the actual rate of anaphylaxis, at least in this study cohort, was up to 25-100 times higher.
A Harvard studyiv has shown that less than 1% of AEs are reported in VAERS. This UK Gov. webpagev on the UK ‘Yellow Card’ reporting system states the reporting rate is 10% for serious AEs and 2-4% for minor AEs. In NZ the reporting rate for AEs on CARM is estimated at 5%vi of the actual incidence.
With the rollout of Covid vaccines, the CDC in the US recognised the failings of the VAERS system and introduced a Covid Vaccine specific, smartphone app called V-Safe. After 6 monthsvii there were nearly 8 million people enrolled in V-Safe. Overall 4 million reported systemic side effects after each dose (7.5million in total). For Pfizer recipients this was 51.3% of first dose recipients and 66.1% of 2nd dose recipients. This roughly confirms the rates of systemic reactions reported in Pfizer’s trial. According to V-safe 20% of Pfizer recipients were not able to carry out normal activities after dose 2 and 12.3% had to take time off work. Over the same time period, VAERS was only reporting 340,000 adverse events in total. Having 2 reporting systems side by side means that VAERS is likely to be even more under reported than it normally would because anyone reporting to V-safe is not likely to report it on VAERS as well.
What all this means is that while it is always true that this or that individual report may not be related to the Vaccine (correlation does not = causation), the overall, real-life, figure is likely to be at least 10-100x higher than the figure stated in the reporting system.
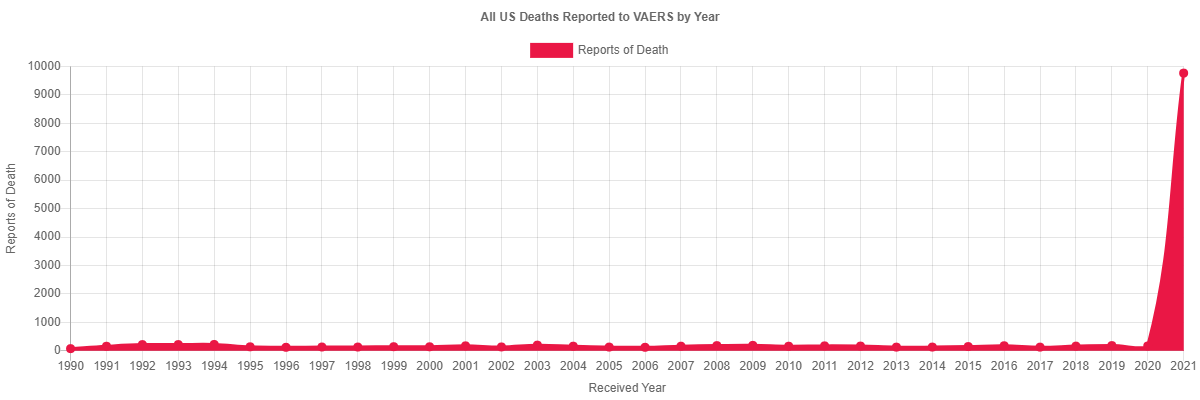
Reporting systems are inaccurate due to under-reporting. But they are good for raising alarms. Here is chart of deaths reported to VAERS from all Vaccines in the US since VAERS started. Look at the number of deaths in 2021 compared to every other year and tell me if you don’t see cause for at least further investigation.
Source COVID Vaccine Data (openvaers.com)viii
What is interesting to see is that government sources hare happy to quote low incidence rates reported to CARM or VAERS etc when it suits them in an effort to pretend there are low incidence rates. Medsafe has done thisix with myocarditis, but without taking into account the known low reporting rate: “they leave this crucial piece of context out:” But when the rates seen in these systems are alarming, such as the death rates shown above, they dismiss the systems as inaccurate. You can’t have it both ways folks!
And, do ‘experts’ really investigate these reports? As of a few months ago there was only one coroners report published in to the death of the 6 or so thousand reported on VAERS.
In summary, databases such as CARM here in NZ or VAERS in the US have a purpose. That purpose is not to be an accurate count of adverse events to vaccines, but instead, to provide a signal to possible safety concerns. The history of these databases around the world has indicated that the actual number of reactions has always been far higher than those reported, but because the inaccuracies of these databases apply to all vaccines, comparisons are indeed a valid way to indicate the presence of possible safety issues. If that comparison shows a rate of reporting far higher than all other vaccines combined, as the VAERS graph above shows, then that is a screaming safety signal that can’t be dismissed just because it does not align with our personal views on vaccine safety. I’m sure that if the databases showed no increase in reporting, if there were no massive spikes following the roll out of C-19 jabs, then Stuff would be all over it promoting VAERS and CARM etc as categoric proof of the safety of the jab. But the opposite is the reality, hence the attempt to dismiss the evidence. All this proves is the bias of the writer.
i https://www.bmj.com/rapid-response/2011/11/02/underreporting-vaccine-adverse-events
ii https://www.bmj.com/content/340/bmj.c2994.full
iii https://jamanetwork.com/journals/jama/fullarticle/2777417
iv https://www.nvic.org/CMSTemplates/NVIC/Pdf/FDA/ahrq-vaers-report-2011.pdf
v https://www.gov.uk/drug-safety-update/yellow-card-please-help-to-reverse-the-decline-in-reporting-of-suspected-adverse-drug-reactions
vi https://www.medsafe.govt.nz/Profs/PUarticles/ADRreport.htm
vii https://www.medrxiv.org/content/10.1101/2021.10.26.21265261v1.full-text
viii https://openvaers.com/covid-data
ix https://www.medsafe.govt.nz/safety/Alerts/comirnaty-myocarditis-alert.htm

CARM ID - AEFI-A-049633
Nov 2021
10 to 19 Female [12]
Dose1
Anaphylactic reaction (10002198), Asthma (10003553)
Injected 30 SEP 21
Died 05 OCT 21 [5]
VAERS ID - 2089593
https://medalerts.org/vaersdb/findfield.php?IDNUMBER=2089593&WAYBACKHISTORY=ON
CDC 'Split Type': NZPFIZER
INC202101759618
Write-up: Sudden death;
This is a spontaneous report received from a contactable reporter(s) (Consumer or other non HCP) from Regulatory Authority. Regulatory number: AEFI-A-049633 (AEFI Case Number). A 12 year-old female patient received bnt162b2 (COMIRNATY), administration date 30Sep2021 (Batch/Lot number: unknown) as dose 1, single for covid-19 immunisation.
The patient''s relevant medical history and concomitant medications were not reported. The following information was reported: DEATH (death) with onset 05Oct2021 13:00, outcome "fatal", described as "Sudden death". The patient date of death was 05Oct2021.
The reported cause of death was "Sudden death". It was not reported if an autopsy was performed. Clinical course: Sudden death due to unknown causes. No follow-up attempts are possible; information about lot/batch number cannot be obtained. No further information is expected.;
Reported Cause(s) of Death: Sudden death
This article should be published in the NZ media, . It would resonate with a lot of people who have seen, first hand, the link between the pathogen, that is, the mRNA experimental gene therapy, and the suffering caused to themselves and their acquaintances.